Part B 101: Three charts show the program helps control costs for health care system and enrollees
Learn more about the Medicare Part B program.

Part B 101: Three charts show the program helps control costs for health care system and enrollees
Learn more about the Medicare Part B program.

Part B 101: Three charts show the program helps control costs for health care system and enrollees
Medicare Part B covers a wide range of health care services, including physician office visits, hospital outpatient care and medical equipment. It also covers medicines that are usually administered by a physician, like many medicines administered by injections and infusions. Many of the medicines covered by Part B are for serious and complex conditions, such as cancer, rheumatoid arthritis and mental illness, among others.
Over the years, some have raised concerns with Part B reimbursement, specifically the metric known as the average sales price (ASP). However, evidence shows the ASP system and other characteristics unique to Part B are helping to control costs for our health care system and Part B enrollees. Here are three facts about Part B you may not know.
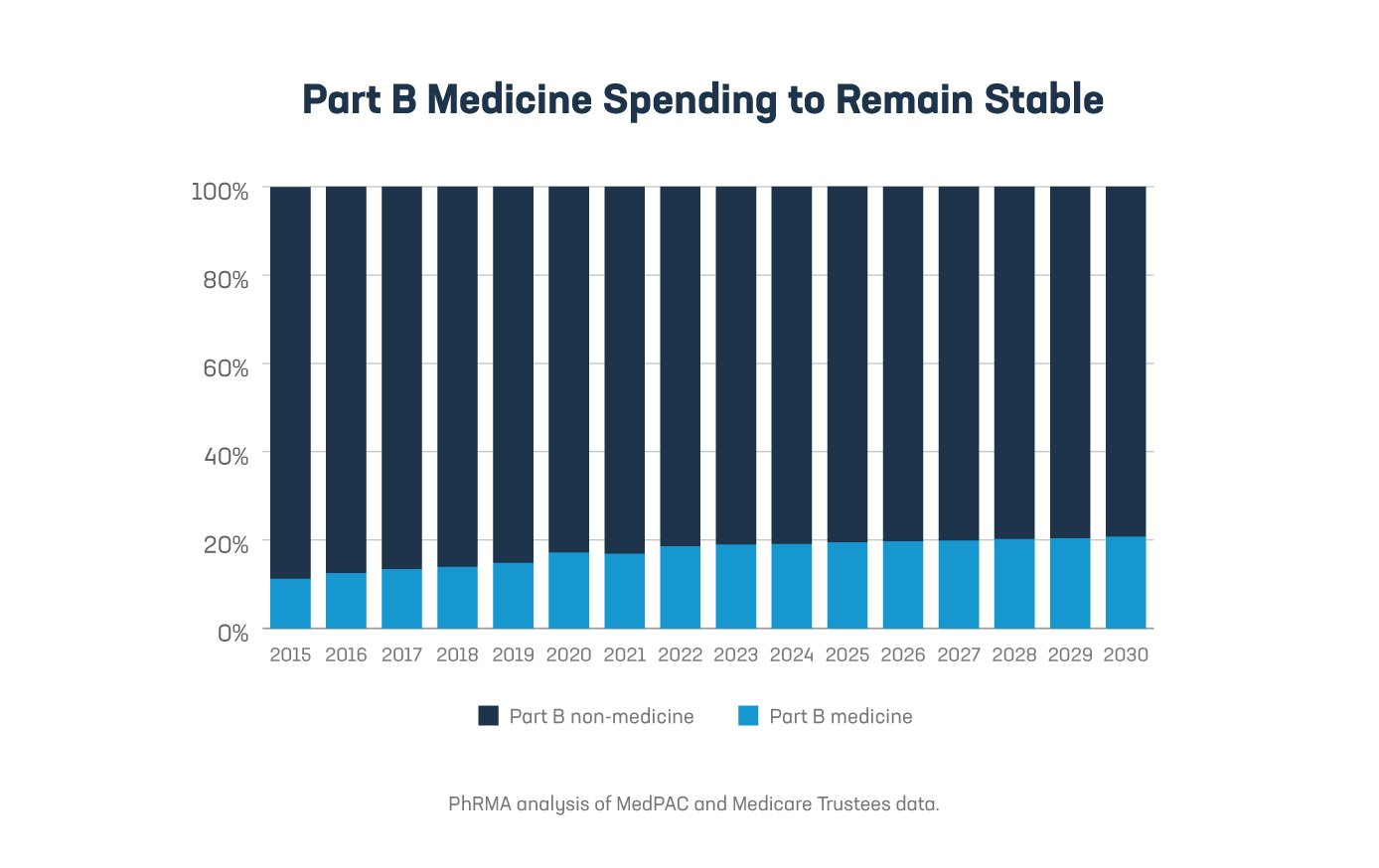
1. Part B medicines are less than 5% of total government Medicare spending. In 2019, of the $787 billion in Medicare spending, only 2% was on outpatient hospital medicines and only 3% was on physician and supplier medicines. This is helped, in part, because reimbursement for Part B medicines remains a stable share of all Part B spending. This is projected to be the case through 2030.
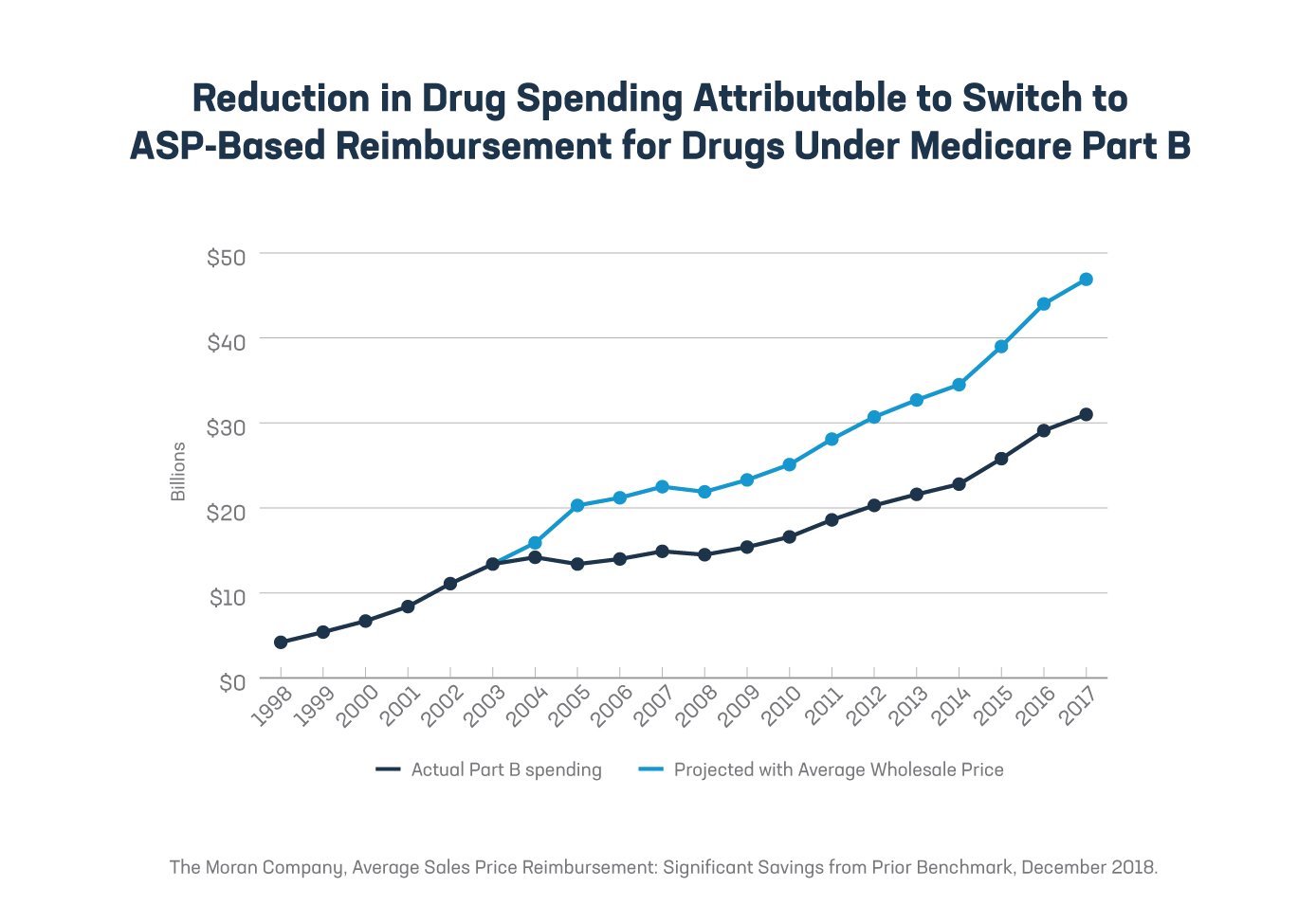
2. The Part B program’s ASP system has saved the government and beneficiaries money. ASP is a market-based price that reflects the average of almost all manufacturer net sales prices and includes most rebates and discounts privately negotiated between manufacturers and payers (with the exception of certain federal and Medicaid discounts and rebates). Congress created the ASP system in 2003 and required ASP-based reimbursement for most Part B medicines starting in 2005. Since that change, we’ve seen lower medicine spending in Part B compared to the previous Part B reimbursement formula (95% of average wholesale price). These savings help contain premiums, coinsurance and deductibles for people enrolled in Medicare. In fact, while medical inflation has been increasing since 2006, volume-weighted ASP for Part B medicines has grown at a slower rate than medical inflation.
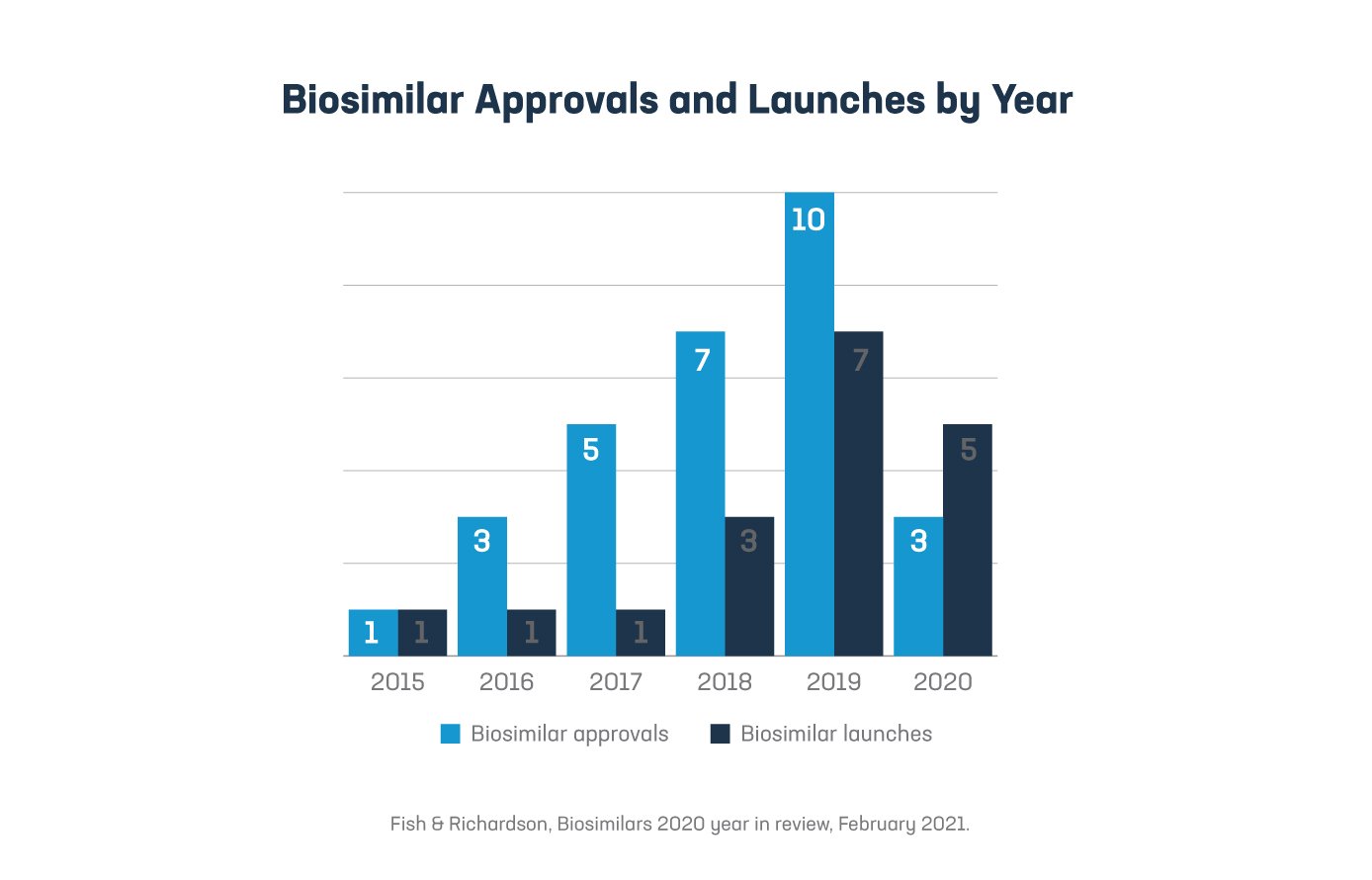
3. An increasing uptake in the use of biosimilars also contributes to growing savings for the program. From 2015 to 2020, the FDA has approved an increasing number of biosimilars for physician-administered medicines. A biosimilar is a type of biologic, which is a medicine made from living organisms through highly complex manufacturing processes. Biologics are at the forefront of scientific and biomedical research and may be able to treat various diseases that do not have other treatments available. According to an IQVIA report, physician-administered biosimilars are anticipated to save more than $100 billion in aggregate over the next 5 years for the Part B program.
While the program is successful in controlling costs for our health care system and Part B enrollees, PhRMA supports ways to modernize how Medicare covers and pays for medicines. This can include a market-based adjustment in Part B that would allow the government and seniors to benefit from more of the savings already negotiated in the commercial market, which could save some seniors hundreds – if not thousands – of dollars each year.